Benzopyrans are selective estrogen receptor Beta agonists with novel activity in models of benign prostatic hyperplasia.
Norman, B.H., Dodge, J.A., Richardson, T.I., Borromeo, P.S., Lugar, C.W., Jones, S.A., Chen, K., Wang, Y., Durst, G.L., Barr, R.J., Montrose-Rafizadeh, C., Osborne, H.E., Amos, R.M., Guo, S., Boodhoo, A., Krishnan, V.(2006) J Med Chem 49: 6155-6157
- PubMed: 17034120
- DOI: https://doi.org/10.1021/jm060491j
- Primary Citation of Related Structures:
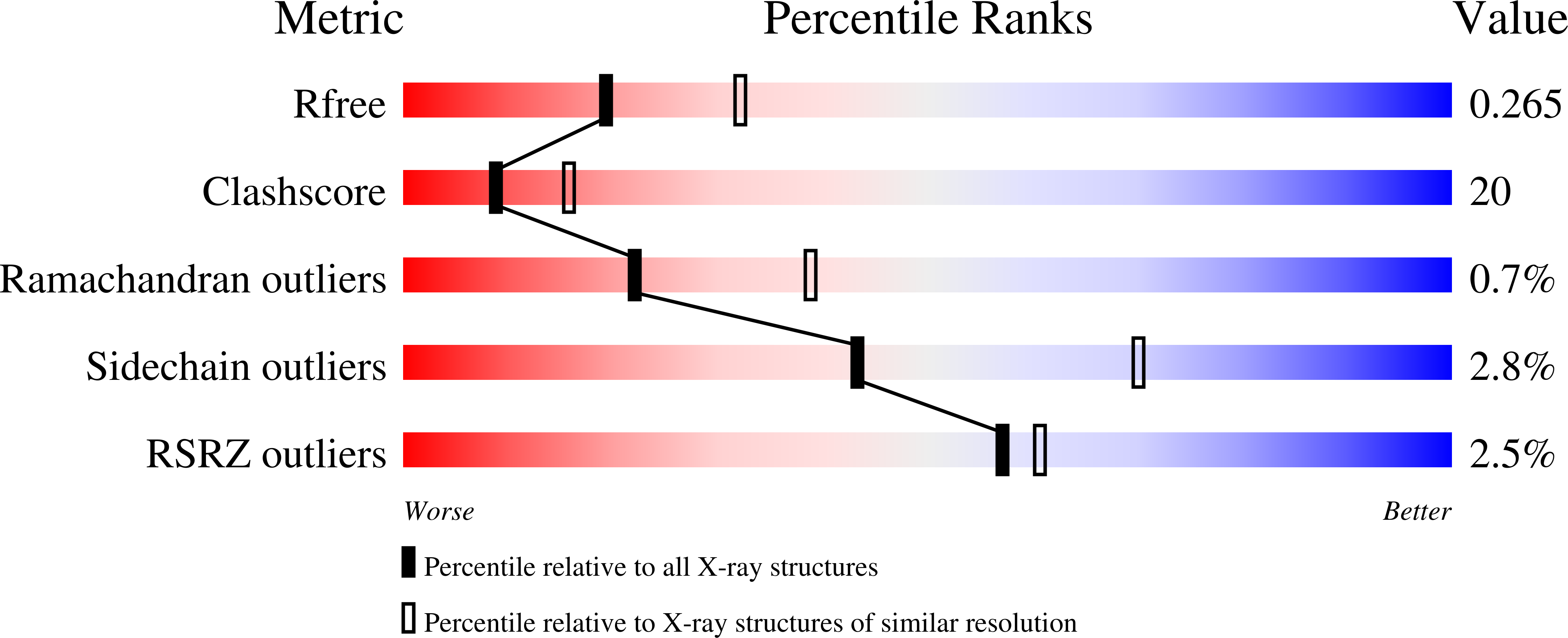
2I0G, 2I0J - PubMed Abstract:
Benzopyran selective estrogen receptor beta agonist-1 (SERBA-1) shows potent, selective binding and agonist function in estrogen receptor beta (ERbeta) in vitro assays. X-ray crystal structures of SERBA-1 in ERalpha and beta help explain observed beta-selectivity of this ligand. SERBA-1 in vivo demonstrates involution of the ventral prostate in CD-1 mice (ERbeta effect), while having no effect on gonadal hormone levels (ERalpha effect) at 10x the efficacious dose, consistent with in vitro properties of this molecule.
Organizational Affiliation:
Discovery Chemistry Research, Bone and Inflammation Research, Lead Optimization Biology, Eli Lilly and Company, Lilly Corporate Center, Indianapolis, Indiana 46285, USA. norman@lilly.com